Energy Exploration Pages:
 Photosynthesis Intro Page Photosynthesis Intro Page
 Energy Flows Through Life Energy Flows Through Life
Other FT Exploring Pages
 Dr Galapagos Dr Galapagos
 What is an Engineer, Really What is an Engineer, Really
 Site Map Site Map
 Link Pages Link Pages
 Lesson Plans Lesson Plans
 Contact Us Contact Us
|
|
|
The First Law of Thermodynamics with Examples
Our changeable friend energy, with its multiple "personalities", is sometimes hard to keep track of and even harder to define. But it does always behave in predictable ways. This means that if you know the rules you, like engineers and scientists, can often predict what will happen and what won't happen.
Smart scientists and engineers, through clever experiments and even cleverer thinking, figured them out in days gone by. It took some open-minded creative thinking. It was not obvious to anyone back then. And much of what was obvious back then about energy was wrong.
|
This section is on the First Law of Thermodynamics, or the Conservation of Energy as it is often called. To start, let's remind ourselves of a few things.
A Few things to remember:
Energy can be stored.
Energy can move from one bunch or piece of matter to another bunch or piece of matter.
Energy can be transformed from one type of energy to another type of energy. |
|
|
|
 |
|
|
|
|
|
A wind turbine converts some, not all, of the kinetic energy in the wind into mechanical and electrical energy. The First Law of Thermodynamics says the sum of the energy put into the wind turbine plus the remaining energy in the air after it passes through the turbine, must exactly equal the energy in the wind before it entered the turbine. |
|
 |
 |
 |
 |
 |
 |
 |
The First Law:
During all this moving and transforming the total amount of energy never changes.
That's it! I've done it!
That's the first law of thermodynamics! Ta da!
Energy changes form and moves from place to place but the total amount doesn't change.
Simple, eh?
 Other ways to say it: Other ways to say it:
It's conservation of energy. Energy is conserved during any and every process or transformation or "happening".
Energy doesn't pop into existence from nowhere.
Energy doesn't pop out of existence into nowhere.
Energy is neither created nor destroyed (the old fuddy duddy way of saying it).
"Energy in" equals "energy stored" plus "energy out"
(if no energy is stored, then "Energy in" equals "Energy out").
You may have already heard that the 1st Law says energy is "always conserved". So why are we always saying we need to conserve more energy if it is "always conserved" already? Darn old english language, same word but different meanings. When we say energy is always conserved we mean the total amount of energy in any process or reaction never changes. When we tell ourselves to conserve energy we mean to use less of it by doing things like driving more fuel efficient cars, or insulating our houses better.
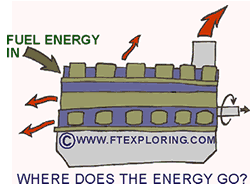 But hold on, if energy never goes away, if the total amount always stays the same, why do we have to worry about not wasting it? Can't we just keep reusing it? We don't really "use" energy, we convert it, and it never "get's used up". But hold on, if energy never goes away, if the total amount always stays the same, why do we have to worry about not wasting it? Can't we just keep reusing it? We don't really "use" energy, we convert it, and it never "get's used up".
It's not a stupid question. It's a very good question. Unfortunately, the First Law of Thermo doesn't really answer it. The popular first law just says the total amount of energy doesn't change.
Scientists and Engineers had to discover a second law of thermodynamics to answer questions like those above. This section is only on the first law but you can jump to the Second Law page now for a very good explanation of the gist of that law, or continue on to the fascinating next page for some good examples to further clarify the First Law. I hope you will read all the pages, for the sake of my fragile ego.
 On page two of this section we'll give some examples of the results of the conservation of energy. Click on the Next Page link below. On page two of this section we'll give some examples of the results of the conservation of energy. Click on the Next Page link below.
|
|
|
|
|
|
<< Previous Page The First Law of Thermodynamics Next Page >>
|
|
|
The Second Law of Thermodynamics |
|
 |
 |
 |
|
|
| |



The LINKS above and below are
NOT paid ads.
They are part of my
ACh Lift projects and interests.
Enjoy them:

Visit my porch

|
 |
 |
|
|
©Copyright 2005, 2007, 2014 David Watson. All rights reserved. Everything in the FT Exploring web site is copyrighted, either by us or by someone else. For information concerning use of this material, click on the word Copyright to go to our legal page. |
|
|
|
|
|
|
|
|
Digressions & Further Explanations Section
(Late breaking news: The Second Law pages are finally here!!)
Energy is Conserved but Not Its Usefulness
As we said above, the first law of thermodynamics says the total amount of energy never changes, even after many changes. It doesn't answer questions like "why can't we re-use the energy over and over again?" or "where do my son's guitar picks go?"
Alas, the second law does answer the questions about re-using energy (not the one's about missing guitar picks and socks though, those mysteries are still beyond the realm of science).
The Second Law is not as easy to say as the first law. It can't be described in one or two sentences. We can't cover it all here, but we can give a brief answer to the questions above. The second law deals with the availability or useability of energy. The unhappy truth is that as energy gets used or transformed in machines or living animals it gets converted into a form that is less useful than before. Something in the energy does get "used up" or even "destroyed". That something is what you might call "usefulness" or as it is often called in thermodynamics, "availability". The energy becomes less available for us to use.
In engines, like those in cars, trucks, locomotives, and airplanes, the energy in the fuel is highly concentrated, meaning there is a lot of energy in a fairly small light package. Each gallon of gasoline (petrol for those of you in other lands) or diesel fuel or jet fuel contains a lot of concentrated easy to use energy. Once that fuel is burned in the engines we are all so dependant on, it gets converted into highly concentrated thermal energy (high temperature) and mechanical energy. The mechanical energy is used to do useful work for us like moving people or freight from one place to another. But eventually all of the fuel's concentrated useful energy get's changed into "low grade" thermal energy. Most is converted immediately into "heat" and goes out of the engine in the cooling water or the engine's hot exhaust gas. The useful mechanical energy gets turned into heat through friction of moving parts or by pushing air molecules out of the way. The molecules that get pushed out of the way absorb the energy of the car or train as increased kinetic energy of the air molecules.
All of the fuel's formerly concentrated "high grade" energy, becomes what we engineer's call "low grade" energy or "waste" heat. We call it that because it is no longer useful to us. It's there, but it's sort of messy and all spread out now, to "gather it up" would take more energy than it's worth. To reuse that low grade energy is simply impossible or would be too expensive and impractical to contemplate. So there it goes, all absorbed into the air and the objects around us as low temperature low grade energy which will finally radiate into outer space to help warm up the universe a tiny little bit. We have to replenish it by burning more fuel. If the fuel is what we call fossil fuel it will eventually all be gone, along with all of its cheap easy to use energy.
Since we haven't yet figured out how to use other forms of energy or alternative fuels as well yet, and those alternatives are likely to be more expensive, most of us think it should not be wasted. That's why we talk about the need to conserve energy. The first law tells us that energy is forever and the total amount stays the same. The second law tells us that "high grade" useful energy always gets turned into "low grade" less useful energy that is difficult or impossible to re-use.
The same is true of the energy conversions in living organisms. The food you eat eventually gets converted into heat (thermal energy) and mechanical work. The heat is useful for a little while to help keep your body temperature constant. The mechanical work is used to make more cells, to breathe, think, run, grow, etc. But in the end, just like with our human made machines, all of the concentrated high grade food energy gets converted into low grade energy we can't reuse. All of it eventually ends up as thermal energy (often called heat) spread out into our surroundings. This also ends up getting radiated into space. And, as you know if you've read the photosynthesis pages, we would run out of useful energy for life very quickly if it didn't get replenished every day by a fresh dose of concentrated energy from the sun.
Back to Where You Were |
|
|
|
|
|
|
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |

