Energy
Exploration Pages:
 Photosynthesis!
Photosynthesis!
 Energy Flows Through Life
Energy Flows Through Life
Other FT Exploring Sections
 Solar Energy!
Solar Energy!
 The Definition of Life!
The Definition of Life!
 Biology & Cell Metabolism!
Biology & Cell Metabolism!
 What
is Engineering, Really? What
is Engineering, Really?
 Dr
Galapagos Dr
Galapagos
 Site Map
Site Map
 Link
Pages Link
Pages
 Contact Me
Contact Me
|
|
| |
Examples
of The Second Law of Thermodynamics
or How
Energy Flows from Useful to Not-So Useful
The
Unstoppable Tendency of Energy
We've said it often in this website:
Everything that happens is caused by an energy
change. Energy changes form, or moves from place to place.
Energy changes are the driving force
of the universe. The driving force of all energy change is the unstoppable tendency
of energy to flow from high concentrations of energy to lower concentrations of
energy.
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
| |
|

|
|
 |
 |
 |
 |
| |
|
This
athlete is converting concentrated food energy through work processes into mechanical muscle movement, kinetic energy of motion, and thermal
energy (heat) - mostly thermal energy. All of the mechanical energy will be converted
to low-grade thermal energy by the end of the race. |
|
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
| |
|
|
|
 |
 |
 |
 |
| |
|
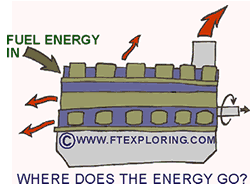
This
diesel engine will convert all of the concentrated fuel energy into work and thermal
energy - mostly thermal energy. Eventually, the mechanical energy will also become
low-grade thermal energy. The total amount of energy hasn't changed, but it is
now so "spread-out" that it cannot be re-used. |
|
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
This is the first
thing to understand about the 2nd Law. Energy always and inevitably flows from
higher concentrations to lower concentrations.
The next thing to understand is that every time energy changes or moves,
some of it, or all of it, becomes less useful. That is the unchangeable result
of becoming less concentrated.
One thing leads to another - always, always, always - cause and effect. Energy
always, and only, goes from more concentrated to less concentrated. Being less
concentrated also means less useful. The cause of everything that happens is a
difference in energy concentration. The effect of every energy change is less
useful energy.
Get it? Every time energy moves, it moves to a condition in which some of it or all of it becomes less useful.
Less Concentrated = Less Useful
Examples
When it comes
to doing useful work like running or powering a car, highly concentrated energy
is easier to use and more efficient than low concentrations of energy.
Foods like carbohydrates and fats, and liquid fuels like gasoline (petrol for
some of you), have highly concentrated potential energy stored in their chemical
bonds. They have a lot of energy in a fairly small space and it is efficient for
us to convert that concentrated energy into useful energy to keep our bodies and
our machines going.
Our Machines Convert Concentrated Energy to Less Useful
"Spread-Out" Energy
For example, when a diesel engine turns a generator, the engine's mechanical energy
is converted into electricity. The electricity is still pretty concentrated, but
not all of the mechanical energy is converted to electricity. Some of the energy
"leaks" away through friction and heat. The rotating generator pushes
air molecules around. The generator wires are heated up by internal friction as
electrons flow through them. The generator cooling fan heats up more air by blowing
it over the generator to keep it cool. All of this heat "spreads out"
into the air around the generator. The energy is still there, but is no longer
useful to us.
A typical generator converts about 90 to 98 percent of the mechanical energy put
into it into concentrated electricity. The other 2 to 10 percent leaks away into
less useful low grade energy.
As the electrical energy flows through the transmission lines to our houses, the
wires are heated by the flowing electrons and more energy is lost as it heats
up the air around the wires. Finally the electrical energy reaches our houses
where it is converted to heat or mechanical energy. The mechanical energy is also
converted to thermal energy through friction. All of the concentrated rotating
mechanical energy put into the generator has been converted to less concentrated
low-grade thermal energy. Ultimately, it will be radiated to space (the ultimate
dilution), gone forever.
Our Bodies Convert Concetrated Energy to Useless Low-Grade
Energy
Animals, plants, and humans, use energy changes to move, grow, catch food, and
make more of themselves. Cellular respiration
is the process by which most living things convert concentrated food energy into
work and thermal energy (often called heat), just as car and truck engines convert
fuel energy into work and thermal energy (still often called heat). Our bodies
do this by controlling complex chemical reactions in which concentrated energy
is carefully moved from higher molecular bond energy levels to lower molecular
bond energies. On the way, some of the energy (not all) is captured by ATP molecules
and used by our cells to do the useful things described above.
When you exercise, some of the food energy gets converted into muscle work, but
most of it gets converted to what we engineers call low-grade thermal energy.
That's why you get all hot and sweaty. In fact, more than 60% of the food energy
is converted to body-warming sweat-making thermal energy during metabolism of
food energy. That leaves only 40% to do useful work in the cells. If you also
figure in the energy required to digest the food and to pump it around in blood
to all the cells, the final number can be significantly less than 40%. That's
about the same as many of our human-made engines.
And, as with human-made machines and devices, all of the mechanical work done
by the cells also ends up as low-grade heat (thermal energy), lost to us forever.
The total amount of energy hasn't changed (1st law), but we can't use it anymore
(2nd Law).
An Example of Energy's Downhill Flow to Uselessness
Let's build a dam.
The dam makes a big lake, with very deep high-pressure water at the base of the
dam. Now we can make a big tube through the dam to guide the high-pressure water
through a big turbine as it flows to the low-pressure side, where the energy is
less concentrated. The turbine converts the high-pressure energy in the water
to mechanical energy, which drives a big generator. The big generator converts
most of the mechanical energy of the turbine into electrical energy. Some, as
we learned above, leaks away to less concentrated low-grade thermal energy.
The electrical energy is still pretty concentrated as it is driven by high voltage
through power lines to our house (it is flowing from higher voltage potential
to lower voltage potential). Along the way a little more concentrated energy "leaks"
into the air around the power lines, becoming low-grade thermal energy. Engineers
call this "transmission losses". It is gone forever.

When the remaining electrical energy reaches our homes, we each take a small portion
and convert it to mechanical energy in a washing machine, or maybe to thermal
energy in an electrical clothes dryer, as well as mechanical energy to tumble
the clothes. All of the mechanical energy in the washer and dryer is eventually
also converted to low-grade thermal energy through friction.
The final destination of all the energy from the hydroelectric power plant is
thousands of light bulbs, electric heaters, toasters, washing machines, electric
dryers, water heaters, stereos, televisions, and other electrical devices in which
it is always and finally converted to low-grade thermal energy that heats up the
air around us a little bit. All that formerly concentrated energy is much less
concentrated now, all spread out in a form we can't re-use. Eventually it radiates
out into cold space as electromagnetic radiation.
What about the water in the dam? That can be replaced right?
Sure. All we need is more fresh (emphasis on fresh) concentrated solar energy
to evaporate the water from the oceans and lakes. Then we need some solar powered
wind energy to blow the clouds over the land where they can form into rain droplets.
Now we need gravity to pull the raindrops to the ground and make the water in
streams and rivers flow downhill to the dam. Finally, we still need gravity to
make the weight of deep water create highly concentrated pressure energy to drive
the turbine.
But all of the formerly fresh concentrated solar energy will once again end up
as low-grade spread out energy that can't be re-used. It too will radiate out
into the frigid cold of space, nevermore to be used. We need continuous fresh
doses of solar energy to keep the cycle going.
That's why we call solar energy renewable. Hydroelectric energy, like wind energy,
is a form of indirect solar energy. It is replenished by the sun.
Petroleum is a fossil fuel. It is not replenished. It is not renewable. When all
of our fossil fuel has been converted to low-grade thermal energy and radiated
into space, that will be the end of it.
The Big Picture Summary
To our human bodies and to our machines, the Second Law describes how energy eventually
and always "runs down" until it can't be re-used for anything except warming the
environment (air, rocks, ground, water) around us. It means, in the big picture,
that the world of living things powered by the sun, would quickly run down to
a cold condition of "not-living", if we didn't get a fresh dose of concentrated
solar energy every day.
And what about non-renewable fuels like coal, petroleum, and natural gas?
Once the potential chemical energy stored in that stuff is converted to mechanical
and thermal energy, it is lost, lost, lost, never to be re-used - forever.
And if you use 250 horsepower more than you need to drive yourself to work, you
have wasted a lot of irreplaceable concentrated energy - turned all of it into
waste heat and flushed it right down the metaphorical toilet.
But there is more to the fascinating story of the Second Law. Scroll down to the
links below to move to the next page.
|
|
| |
|
|
| |
|
|
 |
 |
 |
|
|
|
